Intro to Pharma 4.0 and facility digitalization
Bringing Industry 4.0 to Pharma
The pharmaceutical manufacturing industry is undergoing a digital transformation driven by emerging technologies and the need for increased efficiency and innovation. Known as Pharma 4.0, the fourth industrial revolution in pharmaceutical manufacturing holds immense potential to revolutionize drug development, production, and distribution.
At the core of this transformation is the recognition that data has become the new currency in the pharmaceutical manufacturing landscape. This article explores the importance of data in Pharma 4.0 and its role in reshaping the industry.
If you’d prefer to quickly scan through this article for main points – we added a few bullet points at the end of each major section summarizing the content for your convenience.
The Future is Now: Why Embracing Pharma 4.0 is Imperative for Pharmaceutical Manufacturers
In today’s rapidly evolving landscape, embracing Pharma 4.0 has become a necessity for pharmaceutical manufacturers seeking to thrive in an industry that demands innovation, efficiency, and agility.
By adopting a Pharma 4.0 based approach, pharmaceutical manufacturers can overcome a multitude of manufacturing challenges and position themselves for long-term success.
One of the significant challenges that Pharma 4.0 addresses is the need for enhanced efficiency in manufacturing processes.
Traditional manufacturing methods often rely on manual labor, which can be time-consuming, error-prone, and costly. Pharma 4.0 leverages data, automation, robotics, and advanced analytics to streamline manufacturing operations, reducing production cycle times and increasing overall productivity.
Key Challenges Pharmaceutical Manufacturers Encounter on a Daily Basis, that can be Countered with Pharma 4.0
Maintaining Regulatory Compliance
The pharmaceutical industry is subject to strict regulations and compliance requirements imposed by regulatory bodies such as the FDA (Food and Drug Administration). Manufacturers must also adhere to rigorous standards, such as 21 CFR, to ensure product safety, efficacy, and quality.
Navigating the complex regulatory landscape, staying up to date with changing guidelines, and meeting compliance requirements can be a significant challenge for pharmaceutical manufacturers.
Quality Control
Maintaining consistent product quality is of utmost importance in the pharmaceutical industry.

Manufacturers must implement robust quality control processes to ensure that every batch of medication meets the required standards. This involves comprehensive testing, documentation, and adherence to Good Manufacturing Practices (GMP).
Challenges may arise in areas such as process validation, equipment calibration, and ensuring product stability throughout its shelf life.
It’s a constant challenge for pharmaceutical manufacturers to achieve the “Golden Batch.”
Paper-Based Systems on the Shop Floor

Paper-based systems have long been a challenge for pharmaceutical manufacturers, hindering operational efficiency and compliance. However, the adoption of electronic Batch Records (eBR) offers a transformative solution with significant benefits.
Manual errors, illegible handwriting, and misplaced documents are common pitfalls of paper-based systems. eBR standardizes data entry and automates workflows, ensuring accuracy and data integrity. With digital records, manufacturers can eliminate human errors and provide consistent, complete information.
Searching for and retrieving paper records can be time-consuming, leading to delays and productivity loss.
Embracing eBR in pharmaceutical manufacturing streamlines operations, improves accuracy, and ensures compliance. By transitioning from paper-based systems to digital records, manufacturers can unlock the full potential of their processes.
Rework and Scrap
Re-blend, rework, and scrap are major challenges in pharmaceutical manufacturing, affecting operational efficiency and cost effectiveness.
These processes involve handling materials that do not meet specifications due to quality control issues, formulation errors, or equipment malfunctions. They disrupt production flow, increase costs, and raise concerns about product integrity and regulatory compliance.
The additional time, resources, and complexity involved in rework processes hinder efficiency. Allocating manpower, equipment, and materials to rectify non-conforming products leads to delays and decreased throughput.
These activities also introduce risks such as dosage variability and stability issues. Ensuring product safety and efficacy is crucial to meet regulatory standards and maintain consumer trust.
Downtime
Downtime is a major challenge in the pharmaceutical manufacturing industry, affecting productivity, efficiency, and profitability. When production comes to a halt due to equipment breakdowns, maintenance activities, or unforeseen events, it results in reduced output, delays in meeting customer demands, and potential revenue loss.
Downtime incurs substantial costs, including idle labor, wasted materials, and expenses for repairs or parts replacement, impacting operational expenses and damaging a company’s reputation.

To mitigate downtime challenges, pharmaceutical manufacturers can adopt proactive maintenance strategies. Implementing preventive maintenance programs, regular equipment inspections, and utilizing condition monitoring technologies help identify potential issues before they result in unplanned downtime.
How does Pharma 4.0 Solve These Challenges?
Data
Pharma 4.0 leverages data to address common challenges in the pharmaceutical manufacturing industry. By harnessing the power of data, pharmaceutical manufacturers can overcome obstacles and drive operational excellence.

Data-Driven Decision-Making
One of the key benefits of Pharma 4.0 is data-driven decision making. Through comprehensive data collection and analysis, manufacturers gain valuable insights into their processes, products, and customers. Accurate and timely data enables informed decision making, optimization of production, and ensures quality and compliance throughout the manufacturing process.
By analyzing large datasets, manufacturers can uncover patterns, trends, and correlations that can lead to increased efficiency, reduced downtime, and improved resource allocation.
The Importance of Data in Pharma 4.0
In the era of Pharma 4.0 manufacturing, data has emerged as a critical asset for pharmaceutical companies.
It enables evidence-based decision-making, facilitates predictive analytics, and drives operational efficiency.
Through comprehensive data collection and analysis, companies gain valuable insights into their manufacturing processes, products, and customers. Accurate and timely data allows stakeholders to make informed decisions, optimize production, and ensure quality and regulatory compliance throughout the manufacturing process.
Some Researchers say that Quality Issues Account for around 65% of Drug Shortages.
There’s good reason for manufacturers to take precautions to mitigate these often unnecessary errors.
Data-Driven Decision-Making Empowers Manufacturers to:
- Identify inefficiencies and optimize production schedules.
- Improve quality control processes and reduce defects.
- Enhance supply chain visibility and traceability.
- Streamline regulatory compliance and reporting.
The Role of IoT in Pharma 4.0 Manufacturing
The Internet of Things (IoT) plays a vital role in Pharma 4.0 manufacturing by connecting devices, machines, and processes, thereby enabling real-time data collection and analysis. IoT sensors and devices monitor various parameters, such as temperature, humidity, and pressure, in manufacturing facilities.
This continuous monitoring ensures that environmental conditions are within the specified range, reducing the risk of product failures or deviations. By integrating IoT technology, pharmaceutical manufacturers can improve efficiency, quality control, and overall productivity.

Furthermore, IoT devices can provide valuable insights into equipment performance and maintenance needs. Real-time data from IoT sensors can identify anomalies or deviations, allowing for proactive maintenance and minimizing downtime.
By leveraging IoT data, manufacturers can optimize equipment utilization, streamline maintenance schedules, and improve overall operational efficiency.
IoT in Pharma 4.0 offers Exciting Possibilities, including:
- Real-time monitoring of manufacturing equipment and processes.
- Predictive maintenance to prevent equipment failures and minimize downtime.
- Remote monitoring for improved quality control.
- Enhanced safety measures through the tracking of personnel and assets.
Cloud Computing and Storage in Pharma 4.0 Manufacturing
Pharma 4.0 manufacturing generates vast amounts of data that require efficient storage and management. Cloud computing offers scalable and flexible solutions to handle the ever-increasing data volume in the pharmaceutical industry.
By migrating to the cloud, manufacturers can leverage on-demand computing resources, reduce infrastructure costs, and access data from anywhere at any time. Cloud-based platforms provide secure data storage, enable collaboration, and support advanced analytics.
Cloud computing also facilitates data integration and interoperability across different systems and locations. Manufacturers can centralize data from various sources, such as manufacturing equipment, supply chain partners, and quality control systems, in a cloud-based repository.
This centralized approach allows for a unified view of data and promotes seamless data exchange and analysis. Additionally, cloud-based analytics platforms enable manufacturers to leverage advanced analytics tools and techniques to extract insights from large datasets, empowering data-driven decision-making.
The Benefits of Cloud Computing in Pharma 4.0 include:
- Scalable infrastructure to accommodate growing data needs.
- Improved accessibility and real-time data sharing across multiple locations.
- Enhanced data security and disaster recovery capabilities.
- Cost savings through reduced “technical debt” or IT infrastructure investments.
Artificial Intelligence and Machine Learning in Pharma 4.0 Manufacturing

Artificial intelligence (AI) and machine learning (ML) are transforming pharmaceutical manufacturing by automating processes, analyzing complex data sets, and extracting valuable insights. AI-powered algorithms can predict equipment failures, optimize production schedules, and detect anomalies in real-time.
ML models can identify patterns and correlations in large datasets, enabling personalized medicine, drug discovery, and more precise clinical trials. The integration of AI and ML technologies into Pharma 4.0 manufacturing systems revolutionizes the industry.
In pharmaceutical manufacturing, AI and ML can enhance quality control processes by automating the analysis of large volumes of data. Machine learning algorithms can quickly identify quality issues, deviations, or potential risks, allowing for immediate corrective actions.
Moreover, AI-driven optimization algorithms can fine-tune manufacturing processes, leading to increased efficiency, reduced waste, and improved overall productivity.
AI and ML Applications in Pharma 4.0 Encompass:
- Predictive maintenance to minimize equipment downtime and maintenance costs.
- Intelligent quality control systems for real-time defect detection.
- Accelerated drug discovery and development through data-driven insights.
Blockchain and Data Integrity in Pharma 4.0 Manufacturing
Data integrity and security are paramount in the pharmaceutical industry, given the sensitive nature of information and the potential impact of counterfeit or low-quality drugs. Blockchain technology offers a decentralized and immutable ledger that ensures transparency, traceability, and data integrity.
By leveraging blockchain, pharmaceutical manufacturers can track the entire supply chain and maintain accurate records.
Blockchain technology also enhances trust and collaboration between stakeholders in the pharmaceutical manufacturing ecosystem. Through smart contracts and shared ledgers, manufacturers can establish secure and auditable transactions with suppliers, contract manufacturers, and regulatory authorities.
This level of transparency and traceability minimizes the risk of substandard drugs entering the supply chain and supports compliance with regulatory requirements.
Data Governance, Regulatory Compliance, and Security Challenges in Pharma 4.0 Manufacturing
While the use of data in Pharma 4.0 manufacturing offers numerous benefits, it also presents challenges related to data governance and security. Compliance with regulatory requirements, protection against cybersecurity threats, and maintaining data privacy are critical considerations for companies in the pharmaceutical industry.
Striking a balance between data accessibility and security is crucial to maintaining trust and ensuring data privacy.
To address data governance and security challenges, pharmaceutical manufacturers should implement robust data governance frameworks.
This involves defining data ownership, access controls, and data classification policies. It is essential to establish data governance committees or teams responsible for enforcing data governance policies and ensuring compliance.
Additionally, implementing robust cybersecurity measures, such as encryption, firewalls, and intrusion detection systems, is necessary to protect sensitive manufacturing data from cyber threats.
Addressing Data Governance and Security Challenges Requires:
- Robust data protection measures, including encryption and access controls.
- Compliance with regulations such as 21 CFR and the FDA.
- Regular audits and assessments to identify vulnerabilities and ensure compliance.
- Employee training and awareness programs to foster a security-conscious culture.
Extracting Value from Data in Pharma 4.0 Manufacturing

The true value of data in Pharma 4.0 manufacturing lies in its ability to drive actionable insights and inform decision-making. Data analytics plays a central role in extracting meaningful information from vast datasets.
By leveraging advanced analytics techniques, pharmaceutical manufacturers can optimize manufacturing processes, identify trends, and predict outcomes.
Advanced analytics tools, such as machine learning algorithms and predictive modeling, enable manufacturers to identify inefficiencies, bottlenecks, and areas for improvement in their manufacturing operations.
By analyzing data from multiple sources, including production data, quality control data, and supply chain data, manufacturers can identify patterns and correlations that provide insights into optimizing production schedules, reducing waste, and improving overall efficiency.
Leveraging Data Analytics Enables Manufacturers to:
- Optimize production processes for increased efficiency and reduced costs.
- Identify patterns and correlations to enhance product quality and yield.
- Conduct data-driven research and development for new drug discovery.
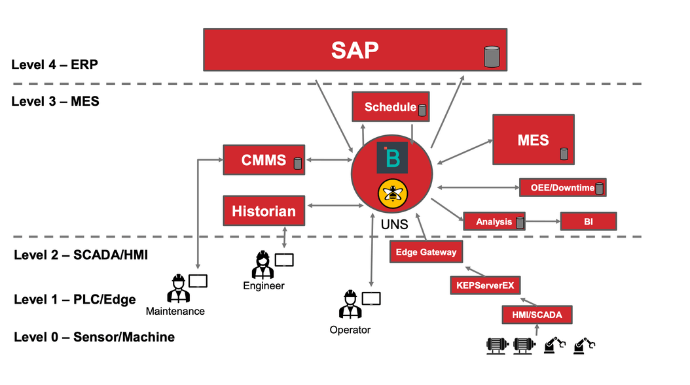
Overcoming Data Silos and Integration Challenges
Pharma 4.0 manufacturing involves the integration of various data sources and systems to create a connected ecosystem. However, many pharmaceutical manufacturers face challenges in breaking down data silos and integrating disparate systems.
Data integration is essential for achieving a holistic view of operations, supply chain, and quality control.
For many years, pharmaceutical companies research and production information have been isolated in “digital islands”
To overcome data silos, manufacturers should adopt interoperable technologies and standardized data formats that facilitate seamless data exchange and integration.
Implementing modern manufacturing execution systems (MES) or manufacturing operations management (MOM) platforms can help centralize data and provide a unified view of manufacturing operations.
Furthermore, investing in application programming interfaces (APIs) and data integration platforms can simplify the integration of different systems, such as enterprise resource planning (ERP), laboratory information management systems (LIMS), and manufacturing equipment.
Strategies to Overcome Data Silos and Integration Challenges include:
- Standardizing data formats and adopting interoperable systems.
- Implementing robust data integration platforms and middleware.
- Creating cross-functional teams to drive data integration initiatives.
- Collaborating with technology partners to leverage integration expertise.
Building a Data-Driven Culture in Pharma 4.0 Manufacturing

To fully realize the potential of data in Pharma 4.0 manufacturing, organizations must cultivate a data-driven culture.
This involves fostering a mindset that values data, investing in training and upskilling the workforce, and creating an environment that encourages experimentation and innovation.
By establishing cross-functional teams and promoting collaboration between data scientists, engineers, and manufacturing professionals, manufacturers can leverage diverse expertise to extract value from data.
Additionally, implementing robust data management systems and collaborating with external partners, such as data analytics firms or technology providers, can bring additional expertise and resources to extract value from data in Pharma 4.0 manufacturing.
Embracing data-driven decision-making throughout the organization, from the shop floor to the executive level, can foster a culture of continuous improvement and innovation.
Building a Data-Driven Culture requires:
- Leadership support and commitment to data-driven decision-making.
- Employee training and upskilling programs to enhance data literacy.
- Establishing data governance frameworks and accountability.
- Encouraging cross-functional collaboration and knowledge sharing.
Conclusion
Future-Proof Operations with Pharma 4.0
Pharma 4.0 is transforming the pharmaceutical manufacturing industry by placing data at the center of manufacturing operations. The integration of advanced technologies such as IoT, cloud computing, AI, ML, and blockchain is unlocking new opportunities for efficiency, quality, and innovation.
However, addressing data governance, security, integration, and cultural challenges is essential for manufacturers to fully harness the power of data in Pharma 4.0.
As pharmaceutical manufacturers continue to navigate the digital technology landscape, embracing data as the new currency will be crucial for driving competitiveness and success.
By leveraging the potential of data-driven decision-making, manufacturers can optimize processes, improve quality control, and deliver the best results possible to their clients or end-users.
The journey to Pharma 4.0 is not without its challenges, but the rewards of enhanced efficiency, improved end-user outcomes, and a competitive edge in the market make it a transformation worth pursuing.